First Line Treatment for Metastatic Pancreatic Adenocarcinoma: Looking for the Step Forward
Abstract
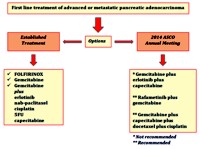
Pancreatic cancer is a lethal disease and its prognosis remains dismal. The modest results of existing available treatments in the first line setting reveal the need of new therapeutic strategies. In this year’s American Society of Clinical Oncology (ASCO) Annual Meeting four remarkable studies were presented regarding this vulnerable group of patients. The efficacy and toxicity profile of gemcitabine plus erlotinib plus capecitabine (Abstract #4122), refametinib plus gemcitabine (Abstract #4025), gemcitabine plus docetaxel plus capecitabine plus cisplatin (Abstract #4135) were examined and the predictive value of a biomarker panel testing response to gemcitabine with or without the addition of erlotinib (Abstract #4133) was also presented.
Image: Therapeutic strategies in first line setting of advanced or metastatic pancreatic adenocarcinoma
Downloads
References
Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA Cancer J Clin. 2014; 64(1):9-29.
SEER Cancer Statistics Review. Surveillance, Epidemiology, and End Results Program, 2013.
Conroy T, Desseigne F, Ychou M, et al. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med. 2011; 364:1817-1825. [PMID:21561347]
Von Hoff DD, Ramanathan RK, Borad MJ, Laheru DA, Smith LS, Wood TE, Korn RL, et al. Gemcitabine Plus nab-Paclitaxel Is an Active Regimen in Patients With Advanced Pancreatic Cancer: A Phase I/II Trial. J Clin Oncol. 2011; 29: 4548-4554. [PMID:21969517]
Berlin JD, Catalano P, Thomas JP, Kugler JW, Haller DG, Benson AB 3rd., et al. Phase III study of gemcitabine in combination with fluorouracil versus gemcitabine alone in patients with advanced pancreatic carcinoma: Eastern Cooperative Oncology Group Trial E2297. J Clin Oncol. 2002; 20:3 270-3275. [PMID:12149301]
Moore MJ, Goldstein D, Hamm J, Figer A, Hecht JR, Gallinger S, Au HJ, et al. Erlotinib plus gemcitabine compared with gemcitabine alone in patients with advanced pancreatic cancer: a phase III trial of the National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol. 2007; 25:1960-1966. [PMID:17452677]
Oliver GR, Sugar E, Laheru D, Diaz LA. Family history of cancer and sensitivity to platinum chemotherapy in pancreatic adenocarcinoma [abstract]. Gastrointestinal Cancers Symposium 2010:180.
Neoptolemos JP, Stocken DD, Friess H, Bassi C, Dunn JA, Hickey H, Beger H, et al. A randomized trial of chemoradiotherapy and chemotherapy after resection of pancreatic cancer. N Engl J Med. 2004; 350: 1200-1210. [PMID:15028824]
Benavides M, Plazas JG, Guillen C, Vera R, Iranzo V, Diaz IA, et al. Gemcitabine(G)/erlotinib(E) versus gemcitabine/ erlotinib/ capecitabine(C) in the first-line treatment of patients with metastatic pancreatic cancer (mPC): Efficacy and safety results of a phase IIb randomized study from the Spanish TTD Collaborative Group. J Clin Oncol. 32:5s, 2014; (suppl; abstr 4122).
Diep CH, Munoz RM, Choudhary A, Von Hoff DD, Han H. Synergistic effect between erlotinib and MEK inhibitors in KRAS wild-type human pancreatic cancer cells. Clin Cancer Res. 2011; 17(9):2744-56. [PMID:21385921]
Van Laethem JL, Jassem J, Heinemann V, Weekes CD, Bridgewater JA, Cascinu S, et al. Phase II study of refametinib (BAY 86-9766), an allosteric dual MEK 1/2 inhibitor, and gemcitabine in patients with unresectable, locally advanced, or metastatic pancreatic cancer. J Clin Oncol. 32:5s, 2014; (suppl; abstr 4025).
Le DT, Laheru DA, Purtell K, Uram JN, Wang H, Lawrence S, et al. A phase 2 trial of low-dose multiagent chemotherapy with gemcitabine, docetaxel, capecitabine, and cisplatin (GTX-C) in subjects with metastatic pancreatic cancer. J Clin Oncol. 32:5s, 2014; (suppl; abstr 4135).
Shultz D, Pai J, Graber MS, Heestand GM, Chang DT, ParulekarWr, et al. A novel biomarker panel examining response to gemcitabine (G) with or without erlotinib (E) for pancreatic cancer (PA) therapy in NCIC clinical trials group PA.3. J Clin Oncol. 32:5s, 2014; (suppl; abstr 4133).
Copyright (c) 2014 Vasilis S Ramfidis, Amanda Psyrri, Kostas N Syrigos, Muhammad Wasif Saif

This work is licensed under a Creative Commons Attribution 4.0 International License.
As a member of Publisher International Linking Association, PILA, iMedPub Group’s JOP follows the Creative Commons Attribution License and Scholars Open Access publishing policies. Journal of the Pancreas is the Council Contributor Member of Council of Science Editors (CSE) and following the CSE slogan Education, Ethics, and Evidence for Editors.